
The synergy between Sanivap and Nocotech for comprehensive, sporicidal and non-toxic bio-disinfection.
Bacillus cereus in maternity wards: a danger under high surveillance
Bacillus cereus is a spore-forming bacterium increasingly recognised as an emerging nosocomial agent, particularly in hospitals.[2] Already well known for its effects in cases of food poisoning, it now poses a growing threat in maternity and neonatal wards, where it can cause serious infections in newborns.
B. cereus has been identified in several powdered infant formula used in maternity wards. [1] Infants are particularly vulnerable, mainly because their immune systems are still very immature.
Conventional protocols are proving insufficient to address this public health risk. The bacteria’s spores are not sufficiently affected by commonly used disinfectants and are known for their heat resistance.
Effective disinfection strategies must eliminate Bacillus cereus and its spores—without introducing new chemical risks in such sensitive environments.
With this in mind, Oxy’Pharm supports hospitals with an integrated and proven approach, combining high-temperature steam bio-cleaning (Sanivap) and automated airborne bio-disinfection (Nocospray). These two sporicidal technologies are non-toxic and perfectly compatible with the requirements of neonatology.
Bacillus cereus, a long-underestimated spore-forming bacterium
Bacillus cereus is a ubiquitous bacterium. Widely found in the general environment, it is present in soil, food, on almost all surfaces and even on human skin. It is also a spore-forming bacterium, capable of producing particularly resistant spores. These enable it to survive for long periods in extreme conditions and resist many conventional disinfection processes. [3]
Bacillus cereus is best known for its role in numerous cases of food poisoning. However, for the first time in 2018, researchers demonstrated its role in nosocomial contamination, both within a single establishment and between several hospitals.[2]
Long considered a simple food contaminant, Bacillus cereus is now identified as an emerging nosocomial agent.
A CONTEXT DEFINED BY EXTREME VULNERABILITY
Bacillus cereus is now identified as the second leading cause of foodborne infections in France, responsible for typical digestive symptoms such as diarrhoea and vomiting. But beyond these mild forms, this bacterium can also cause severe clinical infections of non-food origin, particularly in immunocompromised individuals, the elderly and newborns. In maternity and neonatal wards, infants are physiologically very fragile :
- Their immune systems are still immature.
- Their skin and mucous membranes are particularly permeable.
- The medical devices used in intensive care (probes, catheters, incubators, etc.) increase the number of entry points for pathogens.
In this context, the presence of B. cereus in the hospital environment or in food (particularly powdered infant formula) represents an increased risk of infection with potentially serious consequences.
AN IDENTIFIED SOURCE: POWDERED INFANT FORMULA
Bacillus cereus is currently one of the most frequently detected bacterial contaminants in powdered dairy products, particularly in dehydrated infant formula, with contamination rates reaching up to 26% of samples analysed. It is therefore likely to cause serious infections in infants. [1] In neonatology, where the consumption of powdered infant formula is particularly high, the risk is all the more worrying.

The main environmental reservoirs identified include air ventilation and filtration systems, hospital linen, medical devices and the hands of healthcare staff, all of which are potential vectors for cross-contamination in hospitals.[2]
PARTICULARLY RESISTANT SPORES
Bacillus cereus is capable of producing extremely resistant spores, which enable it to survive in hostile environmental conditions, including in pasteurised or heat-treated products such as powdered milk.[1]
This spore form is resistant to many conventional disinfectants and can also form biofilms, making it even more difficult to eradicate in hospital environments. It is this adaptability that explains its persistent presence in the infant formula industry and its transmissibility via products that are otherwise considered safe.
For effective management, it is therefore essential to adopt disinfection processes that can act on the spores without compromising the safety of newborns.
A BACTERIUM NOW RECOGNISED AS A NOSOCOMIAL AGENT
Bacillus cereus is now recognised as a genuine hospital pathogen capable of causing severe local or systemic infections. In newborns and immunocompromised individuals, documented complications include septicaemia, endocarditis, pneumonia, meningitis, encephalitis and tissue destruction, with mortality rates of up to 10%. These severe forms are exacerbated by factors such as prematurity, surgical wounds, or the presence of catheters and medical devices in intensive care.[1][2]
A study conducted by INRA and ANSES, in collaboration with nine French hospitals, highlighted several cases of intra-hospital cross-contamination, some linked to the same strain detected in different patients and in the environment, sometimes over a period of more than two years. Inter-hospital contamination has also been demonstrated, confirming the ability of B. cereus to persist and circulate throughout hospital environments. These observations form the first documented cases of nosocomial epidemics involving this bacterium. [2]
Monitoring its antibiotic resistance profile is therefore becoming essential. Cases of acquired resistance, particularly to rifampicin, have been identified, complicating therapeutic management.[1] These are all reasons for increased vigilance in maternity and neonatal wards.
A comprehensive, safe and efficient response: the synergy of Sanivap and Nocospray
SANIVAP : THE HIGH-TEMPERATURE STEAM BIO-CLEANING SOLUTION WITH PROVEN EFFECTIVENESS AGAINST SPORES

Developed by Oxy’Pharm, Sanivap is a steam bio-cleaning solution designed for high-demand environments such as maternity wards and neonatal units. It works by diffusing saturated steam at 97°C from the accessory outlet, enabling thorough removal of dirt, biofilms and micro-organisms (bacteria, yeasts, viruses and spores) without the use of chemicals.
The steam acts deeply, leaving no residue and requiring no rinsing, while preserving even the most delicate materials. This is an essential advantage for safely disinfecting surfaces in direct contact with newborns, such as incubator mattresses, cots and changing tables.
Sanivap’s effectiveness is certified by the NF T72-110 standard, which guarantees proven disinfecting action, including on spores. For areas with a high biological risk, the SP540H model combines steam and hydrogen peroxide (H₂O₂), reinforcing the sporicidal action while remaining non-toxic and compatible with materials.
Designed for intensive use (up to 10 hours of use before charging), Sanivap adapts to all configurations thanks to a wide range of accessories, including sanitary nozzles, steam mops, textile brushes and siphon bells. Mounted on an ergonomic trolley, it makes handling easier and improves workplace quality of life for staff.
NOCOSPRAY : AUTOMATED AIRBORNE BIO-DISINFECTION FOR HARD-TO-REACH AREAS
As a natural partner of manual bio-cleaning, Oxy’Pharm’s Nocotech technology is based on automated airborne disinfection – ideal for treating inaccessible corners or complex spaces in healthcare settings. This disinfection is carried out without any human contact required.
Once programmed, the Nocospray diffuses an ultra-fine dry mist (≤ 5 µm) at high speed (80 m/s), distributed evenly in volumes of up to 1,000 m³. It is used with Nocolyse (6% H₂O₂) or Nocolyse One Shot (12%) bio-disinfectants, depending on the target microbial load.
After deployment, the mist breaks down naturally into water and oxygen, leaving no residue on surfaces. Moisture-free, non-corrosive and peracetic acid-free, the solution is perfectly compatible with sensitive equipment, whether medical devices, textiles or electronics.
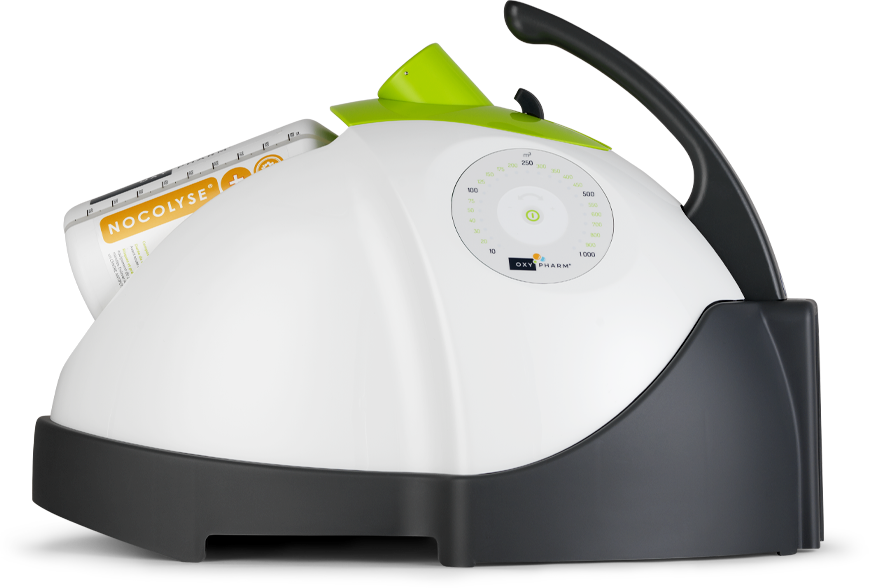
This technology is EN 17272 certified, the leading European standard for automated disinfection processes, with proven effectiveness on spores, bacteria, viruses, yeasts and moulds.
Thanks to its simple programming (volume, dosage, duration, cycles), Nocospray can be easily integrated into hospital protocols without increasing workload for staff. Meanwhile, the more compact Kube model is ideal for effectively treating smaller spaces such as mobile medical units, isolators and airlocks.
Tangible benefits for hospital teams and newborns
Solutions designed for use on the job, ensuring safety, simplicity and ease of use.
OPTIMAL SAFETY FOR THE MOST VULNERABLE PATIENTS
Designed to meet the requirements of critical environments such as neonatology wards, Sanivap and Nocotech solutions provide effective bio-cleaning and scientifically proven bio-disinfection methods against spore-forming or multi-resistant agents, without adding additional toxic load to the environment. They act without residual moisture, chemical residues or aggressive biocides, ensuring complete safety for newborns and staff, sensitive surfaces and medical devices.
SIMPLE TO USE WHILE IMPROVING WORKPLACE QUALITY OF LIFE
Designed with and for professionals in the field, Oxy’Pharm solutions make the work of hygiene and care teams easier. Their ergonomic design (mobile trolleys, accessories adapted to each situation, intuitive programming) makes them easy to use, even for non-technical staff.
Protocols are simpler, faster and better received by staff, as they involve fewer repetitive movements, less physical effort and no rinsing or complex steps. In stressful or emergency situations, this ease of use enables staff to work calmly, without unnecessary pressure. Oxy’Pharm solutions therefore reduce the physical and mental burden on hygiene teams.
Lastly, by drastically limiting exposure to irritating or toxic products, Oxy’Pharm solutions also protect staff health.
The result is better workplace quality of life and more time devoted to protecting the most vulnerable patients.
An eco-friendly approach combining economic efficiency and optimised organisation
Oxy’Pharm solutions have been designed to combine performance with cost and resource management. They significantly reduce the use of water, chemicals and disposable consumables.
The Sanivap & Nocotech synergy is therefore part of an eco-responsible approach to optimising resources and reducing waste, while preserving surfaces and equipment. It also optimises equipment and human resources by ensuring faster cleaning cycles and better room turnover.
Oxy’Pharm’s eco-responsible approach is fully integrated into the quality and sustainable development initiatives of healthcare facilities. It helps to strengthen the image of these facilities among staff, regulatory bodies and families. Faced with the very real risk of Bacillus cereus, Oxy’Pharm is committed to working alongside hospital teams to build a safer, healthier and more respectful environment for everyone.
Thanks to Sanivap and Nocotech, establishments can adopt powerful disinfection protocols against spores, without ever exposing newborns or caregivers to aggressive substances.
At Oxy’Pharm, this is the cornerstone of our approach: no longer having to choose between effectiveness and safety. We want to provide maternity wards with impeccable, reproducible and eco-responsible hygiene, in line with people-centric values.
Sources :
Scientific references: